He also found that the various energy levels can hold differing numbers of electrons: energy level 1 may hold up to 2 electrons, energy level 2 may hold up to 8 electrons, and so on. Nobel Prize (1922) Notable Family Members: son Aage N. This meant that he observed the coloured lines. 1913 was a year of highs and lows in physical sciences. 1 In 1913 Bohr analysed the spectral lines of hydrogen. The events that led to the atomic theory unfolded as a series of scientific breakthroughs and puzzles. Thomsons guidance and In 1912 he was at work in Rutherford’s laboratory in Manchester. Bohr's model calculated the following energies for an electron in the shell, n : E ( n) 1 n 2 13. In 1911 did experimental work going on in the Cavendish Laboratory under J.J. Bohr's model of hydrogen is based on the nonclassical assumption that electrons travel in specific shells, or orbits, around the nucleus. The ground state of the atom was when the shells get closer and closer to one another, the further they are from the nucleus. Niels Bohr was born in Copenhagen on October 7, 1885. He found that the closer an electron is to the nucleus, the less energy it needs, but the farther away it is, the more energy it needs. Bohr called the collection of electrons that orbits at any distance an electron shell. While in their orbit, electrons do not lose any energy. The further they are from the nucleus, the greater amount of energy they possess. But from his special evidence, he concluded that electrons are found at only certain distances from the nucleus, and have particular values of energy. Despite its eventual replacement by more comprehensive models, Bohr’s atomic model holds its place in the history of atomic theory and quantum mechanics. īohr agreed with Rutherford’s proposal that in the atom the electrons revolve around a central positively charged nucleus that is responsible for most of the weight of the atom. Calculations involving radius, energy, velocity, and time period in Bohr’s model provided valuable insights into electron behavior in atoms. Dalton’s theory proposed a number of basic ideas. Dalton’s theory went beyond just a philosophical statement that there are atoms. Bohr’s theory explained the atomic spectrum of hydrogen and established new and broadly applicable principles in quantum mechanics. Dalton’s theory was different from previous discussions of atoms because it had the weight of careful chemical measurements behind it. It was here when he proposed a new atomic model to explain the emission spectrum of hydrogen. John Dalton developed his atomic theory in 1803. 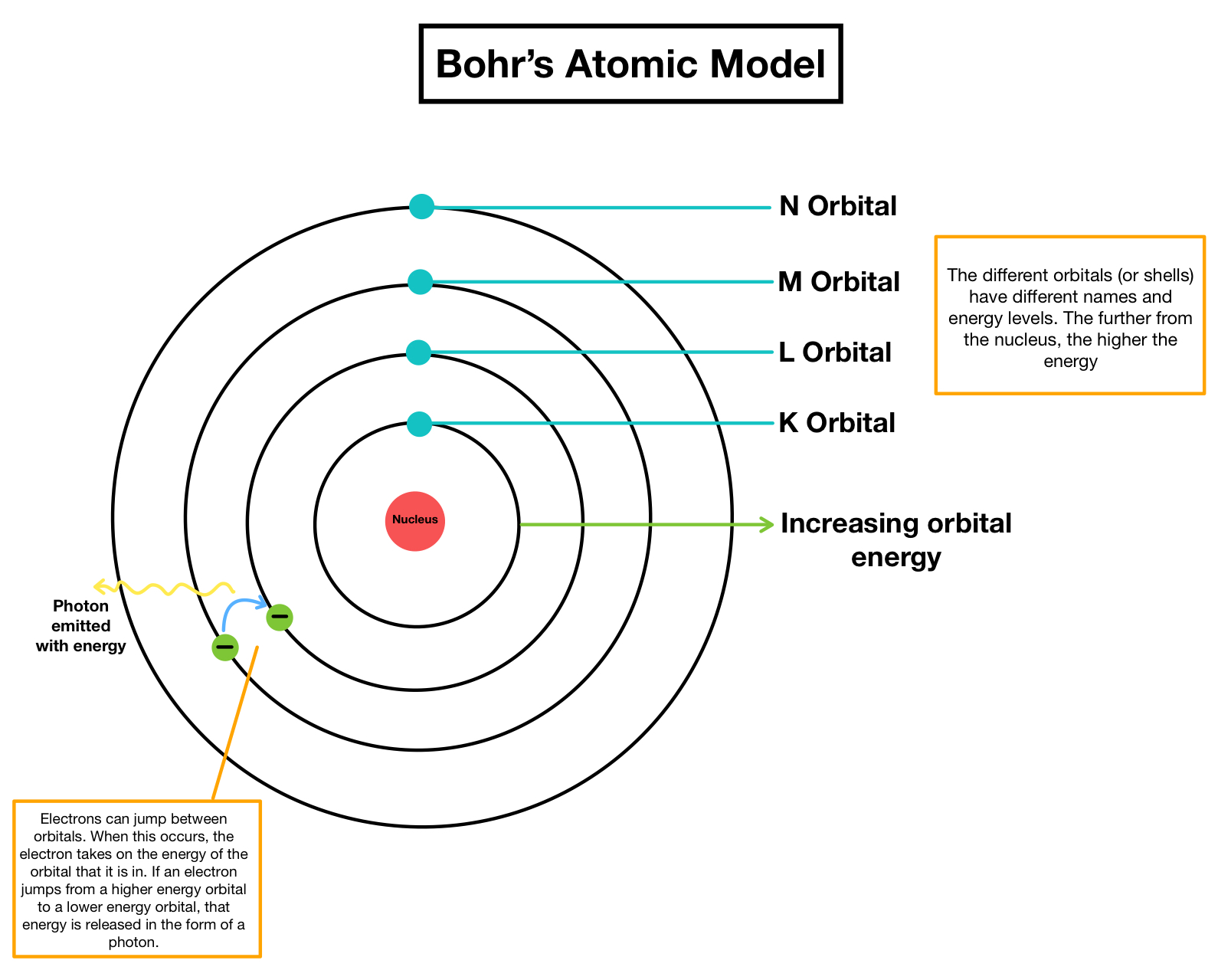
Bohr concluded that the laws of basic physics did not abide to objects on an atomic scale. The new and more general BohrSommerfeld theory described the atom in terms of two quantum numbers, while Bohr had originally used only one quantum number. Since the lines were not continuous like rainbow, the electrons were not giving of energy continuously. Sommerfeld’s extension of the Bohr theory in 19151916, it was turned into a powerful tool of atomic research and adopted and further developed by German physicists in particular. This meant that he observed the coloured lines (wavelengths given by a spectroscope). In 1913 Bohr analysed the spectral lines of hydrogen.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
